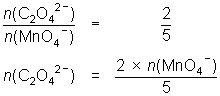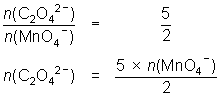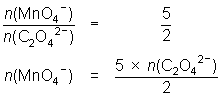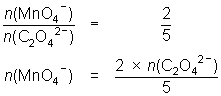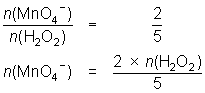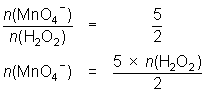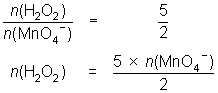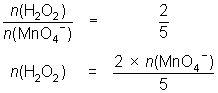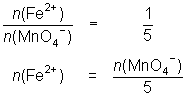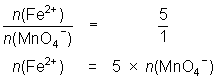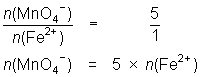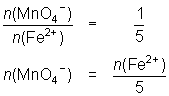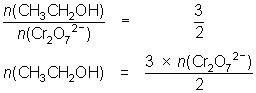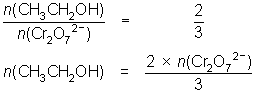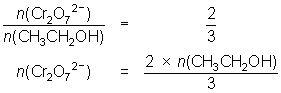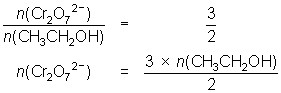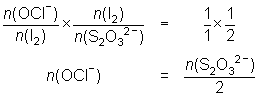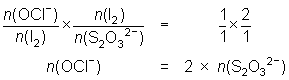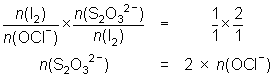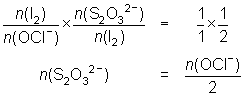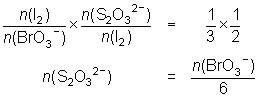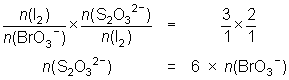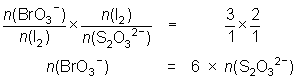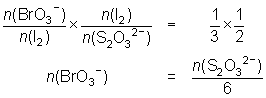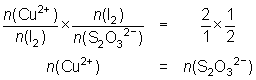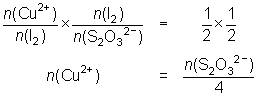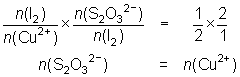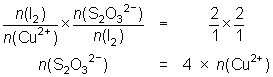20.0 mL of 0.114 mol L-1 oxalic acid reacted with 23.7 mL of MnO4- solution.
5C2O42- + 2MnO4- + 16H+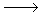 10CO2 + 2Mn2+ + 8H2O
10CO2 + 2Mn2+ + 8H2O-
25.0 mL of diluted hydrogen peroxide solution reacts with 31.1 mL of 0.0184 mol L-1 MnO4- solution.
2MnO4- + 5H2O2 + 6H+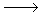 2Mn2+ + 5O2 + 8H2O
2Mn2+ + 5O2 + 8H2O-
13.6 mL of MnO4- solution reacts with 25.0 mL of 0.0848 mol L-1 Fe2+ solution.
MnO4- + 5Fe2+ + 8H+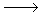 Mn2+ + 5Fe3+ + 4H2O
Mn2+ + 5Fe3+ + 4H2O-
12.3 mL of 0.00400 mol L-1 Cr2O72- reacts with 10.0 mL of ethanol solution.
3CH3CH2OH + 2Cr2O72- + 16H+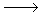 3CH3COOH + 4Cr3+ + 11H2O
3CH3COOH + 4Cr3+ + 11H2O-
20.0 mL of diluted household bleach oxidised KI solution to I2. The iodine was titrated against 0.104 mol L-1 thiosulfate solution and required 32.5 mL.
OCl- + 2H+ + 2I-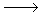 I2 + Cl- + H2O
I2 + Cl- + H2O
I2 + 2S2O32-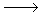 2I- + S4O62-
2I- + S4O62--
20.0 mL of a 0.0175 mol L-1 KBrO3 solution is reacted with KI and the iodine liberated reacted with 28.9 mL of thiosulfate solution.
BrO3- + 6I- + 6H+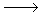 Br- + 3I2 + 3H2O
Br- + 3I2 + 3H2O
I2 + 2S2O32-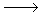 2I- + S4O62-
2I- + S4O62--
10.0 mL of a Cu2+ solution was reacted with KI and the iodine liberated titrated against 0.025 mol L-1 thiosulfate solution. 26.4 mL of thiosulfate were required.
2Cu2+ + 4I-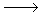 2CuI + I2
2CuI + I2
I2 + 2S2O32-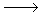 2I- + S4O62-
2I- + S4O62--