
H+(aq), a clear, colourless solution.
Is reduced to...

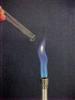
H2(g) A colourless gas which burns with a 'pop'.
Fe2+(aq), a pale green solution.
Is oxidised to...

Fe3+(aq), a pale orange solution which forms a blood-red complex with thiocyanate.
BrO3- (bromate), a clear, colourless solution.
Is reduced to...

Br2(aq). Pure bromine is a red fuming liquid, but in dilute solution it looks orange.

Copper metal is an orange/brown solid.
It is oxidised to...
.jpg)
Cu2+, a blue solution which forms a royal blue complex with ammonia.
Br2(aq), an orange solution, is reduced to...
Br-, a colourless solution.

H+/Cr2O22-, acidified dichromate is orange. It is reduced to...

Cr3+, chromium(III), which is blue-green.
H+/NO3-, concentrated nitric acid, is a clear, colourless solution. It is reduced to...
NO2(g), nitrogen dioxide, a brown gas with a sharp smell.
H+/C2O42-, acidified oxalic acid (which needs to be heated) is a clear, colourless solution. It is oxidised to...
CO2, a colourless gas which turns limewater cloudy.

PbO2, lead dioxide, is a dark brown powder. It is reduced to...


Pb2+, a colourless solution (with a white precipitate) which forms a bright yellow precipitate with iodide.

I-, iodide, is colourless. It is oxidised to ...

I2, iodine, which forms the orange-brown I3- complex in excess I-, and turns starch solution blue-black.
Blue copper sulfate solution (Cu2+) is poured into colourless potassium iodide solution (I-). Name and describe both oxidation and reduction products.
The I- is oxidised to I2, which first turns the iodide orange-brown, and then (as iodide is oxidised) forms a black precipitate. The Cu2+ is reduced to Cu+ which combines with I- to form insoluble, white, CuI.

IO3-, iodate, is colourless. It is reduced to...

I2, iodine, which forms the orange-brown I3- complex in excess I-, and turns starch solution blue-black.

H+/MnO4-, acidified permanganate, is a purple solution. It is reduced to...
Mn2+, which is colourless in dilute solution and very pale pink in concentrated solutions.

MnO4-, neutral permanganate, is a purple solution. It is reduced to...
MnO2(s), manganese dioxide, which is brown.

MnO4-, permanganate, is a purple solution. In alkaline conditions it is reduced to...
MnO42-, which is a dark green solution.
H2O2, hydrogen peroxide, is a colourless liquid. It is oxidised to...
O2(g), oxygen gas, which is colourless (seen here with brown MnO2).
S2O32-, thiosulfate, is a clear, colourless solution. It is oxidised to...

S4O62-, tetrathionate, which is also clear and colourless. (Note: there is no positive test for this ion.)
Cl- in concentrated hydrochloric acid, is a clear, colourless liquid. It is oxidised to...
Cl2(g), a pale green-yellow gas which turns starch-iodide paper blue-black and then bleaches it.
SO32-, sulfite, is colourless. It is oxidised to...
SO42-, sulfate, which is also colourless but forms a white precipitate in acid conditions with barium chloride solution. SO2 gas and HSO3- (hydrogen sulfite, also called bisulfite) are also oxidised to SO42-.
I2, iodine, in excess iodide, is an orange-brown solution. It is reduced to...
I-, iodide, which is colourless.