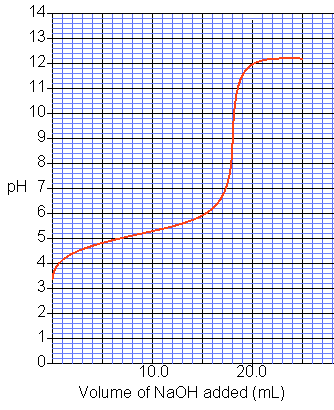
12B 2 Interpreting titration curves
Question one of four.
What volume of NaOH is required to react with all the acid in this titration (1 dp)?
What is the pKa of the acid (to 1 decimal place).
Describe the strength of the acid used in this titration.
Which of the following acid-base indicators should be used for this titration?
- Methyl orange, pKa = 3.7
- Methyl red, pKa = 5.1
- Phenol red, pKa = 7.9
- Thymol blue, pKa = 8.9