|
|
Antarctic sea ice
Ice
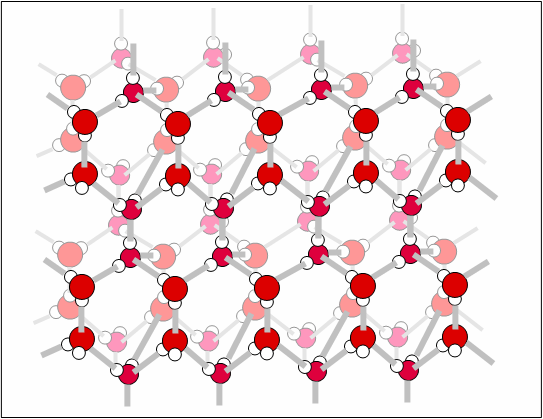
The structure of ice. The grey lines show the hydrogen
bonds between the molecules. Notice that each oxygen atom is connected
to four hydrogen atoms, two covalent bonds within the water molecule, and two hydrogen bonds to adjacent water molecules.
Its lucky for us that hydrogen bonding gives ice its open structure and allows it to float. If water behaved like most other liquids, then as it cooled and solidified, the solid would sink to the bottom. Lakes and seas would freeze from the bottom up. Eventually, the entire ocean would be solid with all the fish inside! Although the sun would melt the surface layer, the warm liquid would float on top and the bottom material would stay frozen. Floating ice, especially the sea ice around Antarctica, is surprisingly important for New Zealanders. IRL scientist Dr Timothy Haskell has spent over 20 years investigating Antarctic sea ice. |
||
|
Why sea ice matters
During the winter months sea ice builds up around the Antarctic land
mass, covering an area of about 19 million km2 thats about 4% of
the total surface area of the Earth and greater than the area of South
America while the summer sea ice area is about 3 million km2 .
Since ice formation is an exothermic process, while melting is endothermic,
these processes absorb or release huge amounts of energy which would
otherwise produce temperature changes in the air and water around Antarctica.
In addition, ice reflects more sunlight that water does, so once the
ice has formed the temperature remains low for longer.
Earths winds are created by the temperature difference between the poles and the equator. Climate scientists need to understand the factors which affect the formation and breaking up of sea ice if they are to make accurate predictions of future weather systems. But sea ice affects more than just our weather systems. Sea water is salty, but sea ice is largely composed of fresh water with the salt left behind in the sea. As the salt concentration in the sea water increases, that water becomes denser and it sinks to the bottom, mixing with the mud, sand and decomposing fish on the sea floor. Its also very cold, and cold water dissolves more oxygen than warmer water. The currents circling Antarctica carry streams of this cold water north, pumping nutrients and oxygen into fisheries of the Pacific and Atlantic oceans. One nutrient-rich stream of water passes up the east coast of New Zealand, attracting the whales to Kaikoura. On the under surface of the sea ice grows algae. Krill feed on this algae, and a large number of fish and sea mammals feed on krill. Marine biologists have discovered that the parts of Antarctica which have a lot of sea ice always have much more fish than those regions where there is little ice. Why we need to know
The sea ice around Antarctica affects climate throughout the world,
and also helps to provide the nutrients for much of the worlds fisheries.
However, these effects are more strongly felt in New Zealand, since we
are relatively close to Antarctica and lie right in the path of one of
those currents of cold water. Thats why New Zealand scientists are studying
all aspects of sea ice, from its structure and strength to the energy
required to melt it and the organisms that live beneath it. What they
learn is used by scientists world-wide to predict the affects of climate
change. Closer to home, the research also helps improve weather forecasting
around New Zealand.
|

Tim and other NZ scientists on a 3 m diameter chunk of sea ice, bobbing up and down in the southern ocean in mid-winter. They are monitoring the energy of the ocean swell that is being removed by the layers of ice as they break and reform.
|
||
|
Tim Haskell
Tim Haskell was raised on a small dairy farm at Opotiki, on the North
Islands East Cape. Tims father had been mechanical engineer before
he took up farming, and he gave Tim had plenty of opportunity to build
things on the farm practical experience that has been very useful in
his varied career.
After gaining his PhD in theoretical physics from Canterbury University, Tim joined the DSIR (Department of Scientific and Industrial Research) in 1972. He worked on solar and industrial heating and was one of the developers of the first nation-wide computer network in New Zealand. He also helped design the machinery that protects some of Wellingtons major buildings (including the Beehive and Te Papa) from a major earthquake. Since 1978 Tim has been a regular visitor to Antarctica. At first, he combined his knowledge of physics with the practical skills gained on the farm to help build roads and runways on the sea ice. Later this work led to the more detailed investigation of the properties of the ice itself. Antarctica is a difficult place to work, presenting unique personal and engineering challenges. Tim is drawn by the wide open spaces, clear air and the need for self-reliance in a place where outside help can be months away.  Tims sea ice camp under a stratospheric mother of pearl cloud in August. The colours are caused by the light from the setting sun being refracted off ice crystals.
|

When he's not on the ice, Tim works in more temperate conditions in Wellington. |
||