World supplies of fossil fuels are falling, and their price is increasing. In response to these problems, many countries are starting to make biofuels from renewable resources such as plants. While the plants grow they remove CO2 from the air to make starch or vegetable oils. These compounds are converted into biofuels such as biodiesel or ethanol. When these fuels are burnt they release CO2 back into the atmosphere, but this process is simply recycling CO2 already in the atmosphere, rather than adding more CO2.
To encourage the use of biofuels, the government is requiring fuel companies to sell minimum amounts of biofuels in New Zealand, starting in April 2008 at 0.53% of total petrol and diesel sold, and rising in following years. Almost certainly these biofuels will be blended at low levels with fossil fuels.
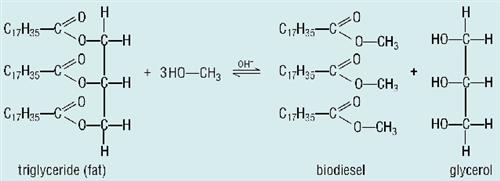
We cant run our trucks on bucket-loads of meat fat: even runny cooking oil is far too thick to use directly in a diesel engine. The fat molecules will need to be broken up first. However, the saponification process used to make soap does not produce a usable fuel because the glycerol makes the mixture sticky. If the glycerol formed is simply removed, the result is a solid fatty acid. Fatty acids make great candles, but you cant run a truck engine on candles.
Carboxylic acids like stearic acid are solid at room temperature because the OH group of one molecule hydrogen bonds with the lone pairs on the C=O of another molecule. If only we could stop them from forming hydrogen bonds.... Easy! Change the OH into an OCH3 .
In biodiesel plants all over the world, chemists are taking fats or oils and reacting them with methanol (or ethanol) in a process called transesterification. The methanol breaks the COO groups in the oils in the same way that water does. The result is a mixture of esters such as methyl stearate, plus glycerol. Because the esters dont form hydrogen bonds, they have lower melting points than their parent acids, so they are liquid at room temperature and they do not mix with the glycerol, so it is a simple matter to drain off the glycerol (which is more dense), leaving the esters behind.
- Most fossil diesel contains sulfur which produces acidic oxides when burnt. Biodiesel contains almost no sulfur.
- Biodiesel is a cleaner-burning fuel, because each molecule contains some oxygen. Quantities of carbon monoxide and particulates (ie particles of carbon) produced are roughly half that produced by fossil diesel. On the down side though, there is about a 10% increase in the amount of nitrogen oxides (which can cause lung damage and acid rain) produced.
- Fossil diesel contains significant amounts of aromatic hydrocarbons. These molecules are thought to cause cancer. Biodiesel contains no aromatic hydrocarbons.
- Because the ester molecules in biodiesel are polar, they are less volatile than the hydrocarbons in fossil diesel, making them less flammable and thus safer to use.
- Biodiesel is also biodegradable in fact, fish will eat it so spills of biodiesel will cause less damage to the environment. Even more impressive: blending biodiesel with fossil diesel makes the fossil diesel break down faster.
The very characteristics that make biodiesel biodegradable and less flammable than hydrocarbon fuels also bring the disadvantages:
- Although esters dont hydrogen bond, they are polar molecules with stronger intermolecular forces than the equivalent hydrocarbons. In low temperatures biodiesel forms a gel which needs to be warmed before use.
- Being polar, the esters in biodiesel are more soluble in water than non-polar hydrocarbons. In theory, if the biodiesel absorbs enough water it will not perform well in an engine. This could, potentially, be a problem with biodiesel used in a marine environment (ferries for example), but trials suggest it is not likely to be an issue on land.
- A biodegradable fuel is great if it spills, but we dont want it to degrade in a fuel tank! Fortunately, the fuel does not degrade as long as it remains in a sealed container.
- Esters are excellent solvents. In particular, they can dissolve away the rubber used in seals and tubing. Modern seals and tubes are made from solvent-resistant polymers, but this issue means that high concentrations of biodiesel cannot be used in older trucks and cars.
At the moment, some New Zealand tallow is used to make soap, and the rest is exported. Converting this low-value by-product into biodiesel for use here makes much more sense than burning fuel to transport it off-shore. If all our tallow were to be converted into biodiesel, it could replace 7.5% of the fossil diesel currently used in New Zealand. 7.5% doesnt seem like a lot, but it would be replacing 150 million litres of imported fossil fuel with a locally grown product made from waste.

Andrew was born in Wales, moved to Australia as a teenager, and came to New Zealand in 1996. He enjoyed the sciences at school, and was keen to find out more about the ideas that interested him. What he did not like was having to study particular processes and formulae for exams. He found the traditional subjects of Chemistry, Physics and Biology too restrictive, so did a degree in Environmental Science at Brisbanes Griffith University . This involved aspects of all the sciences as he studied topics like soils, rocks, natural resources and environmental pollution. It also fitted in nicely with his other passion rock climbing.
After university, Andrew became a professional rock climber. Part of his time was spent in guiding tourists, while for more excitement he worked as an industrial abseiler hanging from ropes off oil rigs, light houses or cliff faces! He was drawn to New Zealand in his hunt for new cliffs to climb and the adventure lifestyle.
One of the ways Andrew used his knowledge of physics, chemistry and rocks was in the design and construction of several large indoor climbing walls. The climbing walls found in many school gymnasiums today are a direct result of Andrews work in the late 90s.
After 10 years climbing, Andrew was ready for a new challenge, so he did a Masters of Applied Science in renewable energy at Massey University in 2001. This lead to his current position at EECA in Wellington.
Many scientists become specialists in ever-narrowing fields of study such as the chemistry of plant sterols. Andrew is a general scientist who, while he does not know a great deal about any one issue, knows how to learn what he needs to know. During those years of rock climbing he worked with a wide variety of people from many countries and many professions. This background helps him to communicate effectively with the many different groups of people he works with today.
Often the research done by scientists doesnt get any further than a report in a journal. Andrew is proud of the work he is doing to make a difference to the way energy is used in New Zealand.

