In 1926 American chemist Irving Langmuir wanted to melt tungsten, which melts at 3407 °C, so he invented a completely different kind of welding torch, the atomic hydrogen torch.
H(g) + H(g) ![]() H2 (g) ΔH = 422 kJ mol 1
H2 (g) ΔH = 422 kJ mol 1
This method releases an incredible 218 kJ g1 of hydrogen. Langmuir could have got more energy per mole of gas used by breaking apart oxygen or nitrogen molecules, but these elements are much heavier than hydrogen. On an energy per gram basis, nothing beats hydrogen. Using his torch he was able to reach temperatures of over 3700 °C.
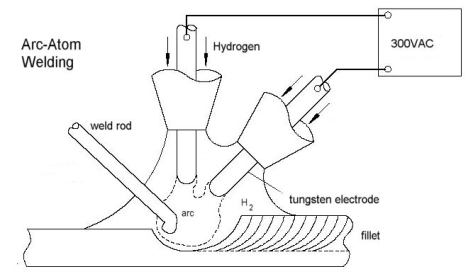
In 1948 a new form of welding was invented gas metal arc welding (GMAW), followed by the similar gas tungsten arc welding (GTAW). In both systems the heat comes from an electric arc while the weld is shielded from the air with some sort of unreactive gas mixtures of helium, argon, nitrogen or sometimes carbon dioxide. In GMAW one electrode is the consumable weld rod, while the other electrode is the metal being welded. In GTAW two tungsten electrodes are used, with the weld rod being added separately (as it is in AHW). These welding systems are much cheaper than AHW, and produce temperatures sufficient to weld steel, aluminium and the other metals commonly in use today.
The solution was obvious: replace air in the bulbs with an unreactive gas. Langmuir used nitrogen, and later bulbs were filled with the even more unreactive argon. Not only did the gas-filled bulbs last longer, but they also used less electricity (the thinner the filament, the greater its resistance). General Electric were very pleased with their young researcher, and gave him the freedom to pursue other interests, whether there was an obvious profit for their company or not.
Langmuir contributed to the work on chemical bonding initiated by Gilbert Lewis. He also investigated the reactions on the surfaces of metals, worked out how to cut down glare on glass surfaces, and was first to try rain making by the method of cloud seeding. He won the Nobel prize in 1932 for his work on surface chemistry.

